Bioactive peptide generation using flavourzyme: functional potentials and emerging applications
DOI:
https://doi.org/10.58951/fstoday.2025.013Keywords:
Hydrolysis. Proteases. Peptides. Bioactivity. Foodtech.Abstract
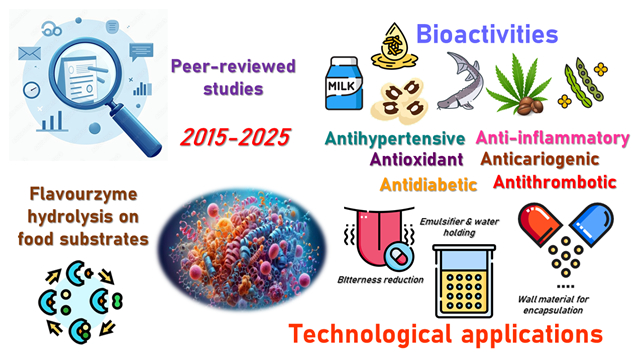
Bioactive peptides derived from enzymatic hydrolysis have attracted considerable attention for their potential use in functional foods to prevent chronic diseases. This review aimed to analyze the functional potential of peptides generated by Flavourzyme-mediated hydrolysis, providing an overview of the scientific evidence on their enzymatic characteristics, substrate specificity, reported biological activities, and emerging applications in food systems. A narrative review approach was applied, focusing on peer-reviewed studies published between 2015 and 2025. Flavourzyme, a protease complex from Aspergillus oryzae, had endo- and exopeptidase activities that facilitate protein degradation and reduce peptide bitterness. The reported bioactivities of its hydrolysates include antioxidant, Angiotensin-Converting Enzyme (ACE) inhibition, dipeptidyl peptidase IV (DPP-IV) inhibition, and anti-inflammatory effects. These activities were observed across diverse protein sources, including dairy, legumes, cereals, and seafood by-products. The enzyme has also been used in sequential hydrolysis strategies with other proteases, improving bioactivity and sensory profiles. Additionally, Flavourzyme applications were identified in technological processes for texture improvement, emulsification, digestibility enhancement, and encapsulation. The results highlighted the enzyme’s versatility and utility in designing functional ingredients with health-promoting effects. This work contributed to identifying the scope and relevance of Flavourzyme in advancing biotechnological strategies for functional food development.
References
Ahmadifard, N., Murueta, J. H. C., Abedian-Kenari, A., Motamedzadegan, A., & Jamali, H. (2016). Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soy-been protein) with SDS-PAGE. Journal of Food Science and Technology, 53(2), 1279–1284. https://doi.org/10.1007/s13197-015-2087-6 DOI: https://doi.org/10.1007/s13197-015-2087-6
Ahtesh, F. B., Stojanovska, L., Mathai, M. L., Apostolopoulos, V., & Mishra, V. K. (2016). Proteolytic and angiotensin‐converting enzyme‐inhibitory activities of selected probiotic bacteria. International Journal of Food Science & Technology, 51(4), 865–874. https://doi.org/10.1111/ijfs.13054 DOI: https://doi.org/10.1111/ijfs.13054
Ahtesh, F. B., Stojanovska, L., Mishra, V., Donkor, O., Feehan, J., Bosevski, M., Mathai, M., & Apostolopoulos, V. (2021). Identification and effects of skim milk-derived bioactive antihypertensive peptides. Biologics, 2(1), 1–14. https://doi.org/10.3390/biologics2010001 DOI: https://doi.org/10.3390/biologics2010001
Ahtesh, F., Stojanovska, L., Shah, N., & Mishra, V. K. (2016). Effect of Flavourzyme ® on angiotensin‐converting enzyme inhibitory peptides formed in skim milk and whey protein concentrate during fermentation by Lactobacillus helveticus. Journal of Food Science, 81(1). https://doi.org/10.1111/1750-3841.13177 DOI: https://doi.org/10.1111/1750-3841.13177
Alahmad, K., Noman, A., Xia, W., Jiang, Q., & Xu, Y. (2023). Influence of the enzymatic hydrolysis using flavourzyme enzyme on functional, secondary structure, and antioxidant characteristics of protein hydrolysates produced from bighead carp (Hypophthalmichthys nobilis). Molecules, 28(2), 519. https://doi.org/10.3390/molecules28020519 DOI: https://doi.org/10.3390/molecules28020519
Asen, N. D., & Aluko, R. E. (2022). Acetylcholinesterase and butyrylcholinesterase inhibitory activities of antioxidant peptides obtained from enzymatic pea protein hydrolysates and their ultrafiltration peptide fractions. Journal of Food Biochemistry, 46(11). https://doi.org/10.1111/jfbc.14289 DOI: https://doi.org/10.1111/jfbc.14289
Bjørlie, M., Hartmann, J. C., Rasmussen, L. H., Yesiltas, B., Sørensen, A.-D. M., Gregersen Echers, S., & Jacobsen, C. (2024). Screening for metal-chelating activity in potato protein hydrolysates using surface plasmon resonance and peptidomics. Antioxidants, 13(3), 346. https://doi.org/10.3390/antiox13030346 DOI: https://doi.org/10.3390/antiox13030346
Cheng, Y.-H., Liu, B.-Q., Cui, B., Wen, L., Xu, Z., Chen, M.-L., & Wu, H. (2023). Alanine substitution to determine the effect of LR5 and YR6 rice peptide structure on antioxidant and anti-inflammatory activity. Nutrients, 15(10), 2373. https://doi.org/10.3390/nu15102373 DOI: https://doi.org/10.3390/nu15102373
Chiang, J. H., Loveday, S. M., Hardacre, A. K., & Parker, M. E. (2019). Effects of enzymatic hydrolysis treatments on the physicochemical properties of beef bone extract using endo‐ and exoproteases. International Journal of Food Science & Technology, 54(1), 111–120. https://doi.org/10.1111/ijfs.13911 DOI: https://doi.org/10.1111/ijfs.13911
Cruz-Chamorro, I., Santos-Sánchez, G., Bollati, C., Bartolomei, M., Li, J., Arnoldi, A., & Lammi, C. (2022). Hempseed (Cannabis sativa) peptides WVSPLAGRT and IGFLIIWV exert anti-inflammatory activity in the LPS-stimulated human hepatic cell line. Journal of Agricultural and Food Chemistry, 70(2), 577–583. https://doi.org/10.1021/acs.jafc.1c07520 DOI: https://doi.org/10.1021/acs.jafc.1c07520
Eberhardt, A., López, E. C., Marino, F., Mammarella, E. J., Manzo, R. M., & Sihufe, G. A. (2021). Whey protein hydrolysis with microbial proteases: Determination of kinetic parameters and bioactive properties for different reaction conditions. International Journal of Dairy Technology, 74(3), 489–504. https://doi.org/10.1111/1471-0307.12795 DOI: https://doi.org/10.1111/1471-0307.12795
Foong, L.-C., Imam, M. U., & Ismail, M. (2015). Iron-binding capacity of defatted rice bran hydrolysate and bioavailability of iron in Caco-2 Cells. Journal of Agricultural and Food Chemistry, 63(41), 9029–9036. https://doi.org/10.1021/acs.jafc.5b03420 DOI: https://doi.org/10.1021/acs.jafc.5b03420
Gerónimo‐Alonso, M., Ortíz‐Vázquez, E., Rodríguez‐Canto, W., Chel‐Guerrero, L., & Betancur‐Ancona, D. (2025). Antithrombotic and anticariogenic activity of peptide fractions from cowpea (Vigna unguiculata) protein hydrolysates. Journal of the Science of Food and Agriculture, 105(1), 209–217. https://doi.org/10.1002/jsfa.13819 DOI: https://doi.org/10.1002/jsfa.13819
Ghulam, A., Arif, M., Unar, A., A. Thafar, M., Albaradei, S., & Worachartcheewan, A. (2025). StackAHTPs: An explainable antihypertensive peptides identifier based on heterogeneous features and stacked learning approach. IET Systems Biology, 19(1). https://doi.org/10.1049/syb2.70002 DOI: https://doi.org/10.1049/syb2.70002
Gui, M., Gao, L., Rao, L., Li, P., Zhang, Y., Han, J., & Li, J. (2022). Bioactive peptides identified from enzymatic hydrolysates of sturgeon skin. Journal of the Science of Food and Agriculture, 102(5), 1948–1957. https://doi.org/10.1002/jsfa.11532 DOI: https://doi.org/10.1002/jsfa.11532
Helal, A., Cattivelli, A., Conte, A., & Tagliazucchi, D. (2023). Effect of ripening and in vitro digestion on bioactive peptides profile in Ras cheese and their biological activities. Biology, 12(7), 948. https://doi.org/10.3390/biology12070948 DOI: https://doi.org/10.3390/biology12070948
Hernandez, L. M. R., & de Mejia, E. G. (2019). Enzymatic production, bioactivity, and bitterness of chickpea (Cicer arietinum) peptides. Comprehensive Reviews in Food Science and Food Safety, 18(6), 1913–1946. https://doi.org/10.1111/1541-4337.12504 DOI: https://doi.org/10.1111/1541-4337.12504
Hunsakul, K., Laokuldilok, T., Sakdatorn, V., Klangpetch, W., Brennan, C. S., & Utama-ang, N. (2022). Optimization of enzymatic hydrolysis by alcalase and flavourzyme to enhance the antioxidant properties of jasmine rice bran protein hydrolysate. Scientific Reports, 12(1), 12582. https://doi.org/10.1038/s41598-022-16821-z DOI: https://doi.org/10.1038/s41598-022-16821-z
Hwang, C., Chen, Y., Luo, C., & Chiang, W. (2016). Antioxidant and antibacterial activities of peptide fractions from flaxseed protein hydrolysed by protease from Bacillus altitudinis HK02. International Journal of Food Science & Technology, 51(3), 681–689. https://doi.org/10.1111/ijfs.13030 DOI: https://doi.org/10.1111/ijfs.13030
Inkanuwat, A., Sukaboon, R., Reamtong, O., Asawanonda, P., Pattaratanakun, A., Saisavoey, T., Sangtanoo, P., & Karnchanatat, A. (2019). Nitric oxide synthesis inhibition and anti-inflammatory effect of polypeptide isolated from chicken feather meal in lipopolysaccharide-stimulated RAW 264.7 macrophages. Food Technology and Biotechnology, 57(2), 200–212. https://doi.org/10.17113/ftb.57.02.19.5964 DOI: https://doi.org/10.17113/ftb.57.02.19.5964
Intiquilla, A., Jiménez‐Aliaga, K., Guzmán, F., Alvarez, C. A., Zavaleta, A. I., Izaguirre, V., & Hernández‐Ledesma, B. (2019). Novel antioxidant peptides obtained by alcalase hydrolysis of Erythrina edulis (pajuro) protein. Journal of the Science of Food and Agriculture, 99(5), 2420–2427. https://doi.org/10.1002/jsfa.9449 DOI: https://doi.org/10.1002/jsfa.9449
Ishikawa, Y., Hira, T., Inoue, D., Harada, Y., Hashimoto, H., Fujii, M., Kadowaki, M., & Hara, H. (2015). Rice protein hydrolysates stimulate GLP-1 secretion, reduce GLP-1 degradation, and lower the glycemic response in rats. Food & Function, 6(8), 2525–2534. https://doi.org/10.1039/C4FO01054J DOI: https://doi.org/10.1039/C4FO01054J
Ji, T., Xu, G., Wu, Y., Wang, Y., Xiao, C., Zhang, B., Xu, B., & Xu, F. (2024). Amelioration of type 2 diabetes mellitus using rapeseed (Brassica napus)-derived peptides through stimulating calcium-sensing receptor: effects on glucagon-like peptide-1 secretion and hepatic lipid metabolism. Journal of Agricultural and Food Chemistry, 72(43), 23804–23818. https://doi.org/10.1021/acs.jafc.4c03987 DOI: https://doi.org/10.1021/acs.jafc.4c03987
Jiang, Y., Li, S., Jiang, L., Mu, G., & Jiang, S. (2025). Immunomodulatory activity and molecular mechanisms of action of peptides derived from casein hydrolysate by alcalase and flavourzyme based on virtual screening. Journal of Dairy Science, 108(3), 2152–2168. https://doi.org/10.3168/jds.2024-25224 DOI: https://doi.org/10.3168/jds.2024-25224
Jogi, N., Mathew, A., & Mamatha, B. S. (2024). Modification of bioactive properties in food protein hydrolysates by alcalase and trypsin. Journal of Health and Allied Sciences NU, 14(S 01), S26–S34. https://doi.org/10.1055/s-0044-1782643 DOI: https://doi.org/10.1055/s-0044-1782643
Karamać, M., Kosińska-Cagnazzo, A., & Kulczyk, A. (2016). Use of different proteases to obtain flaxseed protein hydrolysates with antioxidant activity. International Journal of Molecular Sciences, 17(7), 1027. https://doi.org/10.3390/ijms17071027 DOI: https://doi.org/10.3390/ijms17071027
Karami, Z., Butkinaree, C., Somsong, P., & Duangmal, K. (2023). Assessment of the DPP‐IV inhibitory potential of mung bean and adzuki bean protein hydrolysates using enzymatic hydrolysis process: specificity of peptidases and novel peptides. International Journal of Food Science & Technology, 58(6), 2995–3005. https://doi.org/10.1111/ijfs.16422 DOI: https://doi.org/10.1111/ijfs.16422
Karimi, A., Azizi, M. H., & Ahmadi Gavlighi, H. (2020). Fractionation of hydrolysate from corn germ protein by ultrafiltration: In vitro antidiabetic and antioxidant activity. Food Science & Nutrition, 8(5), 2395–2405. https://doi.org/10.1002/fsn3.1529 DOI: https://doi.org/10.1002/fsn3.1529
Kasiwut, J., Youravong, W., & Sirinupong, N. (2019). Angiotensin I‐converting enzyme inhibitory peptides produced from tuna cooking juice hydrolysate by continuous enzymatic membrane reactor. Journal of Food Biochemistry, 43(12). https://doi.org/10.1111/jfbc.13058 DOI: https://doi.org/10.1111/jfbc.13058
Kim, S. H., Roy, P. K., & Park, S. Y. (2024). Synergistic effects of combined flavourzyme and floating electrode–dielectric barrier discharge plasma on reduction of Escherichia coli biofilms in squid (Todarodes pacificus). Microorganisms, 12(6), 1188. https://doi.org/10.3390/microorganisms12061188 DOI: https://doi.org/10.3390/microorganisms12061188
Lammi, C., Bollati, C., Ferruzza, S., Ranaldi, G., Sambuy, Y., & Arnoldi, A. (2018). Soybean- and lupin-derived peptides inhibit DPP-IV activity on in situ human intestinal Caco-2 cells and ex vivo human serum. Nutrients, 10(8), 1082. https://doi.org/10.3390/nu10081082 DOI: https://doi.org/10.3390/nu10081082
Mao, Y., Chen, L., Zhang, L., Bian, Y., & Meng, C. (2023). Synergistic hydrolysis of soy proteins using immobilized proteases: assessing peptide profiles. Foods, 12(22), 4115. https://doi.org/10.3390/foods12224115 DOI: https://doi.org/10.3390/foods12224115
Matos, F. M. de, Sales, A. C. de A., Tavares, G. M., & Castro, R. J. S. de. (2025). Effects of the Maillard reaction on the antioxidant and antihypertensive properties of black cricket (Gryllus assimilis) protein hydrolysates. ACS Food Science & Technology, 5(4), 1365–1373. https://doi.org/10.1021/acsfoodscitech.4c00947 DOI: https://doi.org/10.1021/acsfoodscitech.4c00947
Mazorra-Manzano, M. A., Ramírez-Suarez, J. C., & Yada, R. Y. (2018). Plant proteases for bioactive peptides release: A review. Critical Reviews in Food Science and Nutrition, 58(13), 2147–2163. https://doi.org/10.1080/10408398.2017.1308312 DOI: https://doi.org/10.1080/10408398.2017.1308312
Merz, M., Eisele, T., Berends, P., Appel, D., Rabe, S., Blank, I., Stressler, T., & Fischer, L. (2015). Flavourzyme, an enzyme preparation with industrial relevance: automated nine-step purification and partial characterization of eight enzymes. Journal of Agricultural and Food Chemistry, 63(23), 5682–5693. https://doi.org/10.1021/acs.jafc.5b01665 DOI: https://doi.org/10.1021/acs.jafc.5b01665
Mirzaee, H., Ahmadi Gavlighi, H., Nikoo, M., Udenigwe, C. C., Rezvankhah, A., & Khodaiyan, F. (2024). Improved antioxidant, antihypertensive, and antidiabetic activities and tailored emulsion stability and foaming properties of mixture of corn gluten and soy protein hydrolysates via enzymatic processing and fractionation. Food Science & Nutrition, 12(11), 9749–9763. https://doi.org/10.1002/fsn3.4532 DOI: https://doi.org/10.1002/fsn3.4532
Mora-Melgem, J. A., Arámburo-Gálvez, J. G., Cárdenas-Torres, F. I., Gonzalez-Santamaria, J., Ramírez-Torres, G. I., Arvizu-Flores, A. A., Figueroa-Salcido, O. G., & Ontiveros, N. (2023). Dipeptidyl peptidase IV inhibitory peptides from chickpea proteins (Cicer arietinum L.): pharmacokinetics, molecular interactions, and multi-bioactivities. Pharmaceuticals, 16(8), 1109. https://doi.org/10.3390/ph16081109 DOI: https://doi.org/10.3390/ph16081109
Naji, A. M., & Çam, M. (2025). Comparative analysis of milk casein hydrolysates from cow, water buffalo, goat and sheep: effects of flavourzyme and neutrase on formation of bioactive peptides. Journal of Food Measurement and Characterization, 19(1), 341–355. https://doi.org/10.1007/s11694-024-02972-2 DOI: https://doi.org/10.1007/s11694-024-02972-2
O’Sullivan, D., Nongonierma, A. B., & FitzGerald, R. J. (2017). Bitterness in sodium caseinate hydrolysates: role of enzyme preparation and degree of hydrolysis. Journal of the Science of Food and Agriculture, 97(13), 4652–4655. https://doi.org/10.1002/jsfa.8306 DOI: https://doi.org/10.1002/jsfa.8306
Oliveira, L. de C., Martinez-Villaluenga, C., Frias, J., Elena Cartea, M., Francisco, M., Cristianini, M., & Peñas, E. (2024). High pressure-assisted enzymatic hydrolysis potentiates the production of quinoa protein hydrolysates with antioxidant and ACE-inhibitory activities. Food Chemistry, 447, 138887. https://doi.org/10.1016/j.foodchem.2024.138887 DOI: https://doi.org/10.1016/j.foodchem.2024.138887
Peighambardoust, S. H., Karami, Z., Pateiro, M., & Lorenzo, J. M. (2021). A review on health-promoting, biological, and functional aspects of bioactive peptides in food applications. Biomolecules, 11(5), 631. https://doi.org/10.3390/biom11050631 DOI: https://doi.org/10.3390/biom11050631
Rezvankhah, A., Yarmand, M. S., Ghanbarzadeh, B., & Mirzaee, H. (2021a). Characterization of bioactive peptides produced from green lentil (Lens culinaris) seed protein concentrate using Alcalase and Flavourzyme in single and sequential hydrolysis. Journal of Food Processing and Preservation, 45(11). https://doi.org/10.1111/jfpp.15932 DOI: https://doi.org/10.1111/jfpp.15932
Rezvankhah, A., Yarmand, M. S., Ghanbarzadeh, B., & Mirzaee, H. (2021b). Generation of bioactive peptides from lentil protein: degree of hydrolysis, antioxidant activity, phenol content, ACE-inhibitory activity, molecular weight, sensory, and functional properties. Journal of Food Measurement and Characterization, 15(6), 5021–5035. https://doi.org/10.1007/s11694-021-01077-4 DOI: https://doi.org/10.1007/s11694-021-01077-4
Rezvankhah, A., Yarmand, M. S., Ghanbarzadeh, B., & Mirzaee, H. (2023). Development of lentil peptides with potent antioxidant, antihypertensive, and antidiabetic activities along with umami taste. Food Science & Nutrition, 11(6), 2974–2989. https://doi.org/10.1002/fsn3.3279 DOI: https://doi.org/10.1002/fsn3.3279
Rocha, T. de S., Hernandez, L. M. R., Mojica, L., Johnson, M. H., Chang, Y. K., & de Mejía, E. G. (2015). Germination of Phaseolus vulgaris and alcalase hydrolysis of its proteins produced bioactive peptides capable of improving markers related to type-2 diabetes in vitro. Food Research International, 76, 150–159. https://doi.org/10.1016/j.foodres.2015.04.041 DOI: https://doi.org/10.1016/j.foodres.2015.04.041
Saetang, J., Haewphet, T., Nilsuwan, K., & Benjakul, S. (2025). ACE- and DPP-IV-inhibitory peptides from Bambara groundnut hydrolysate: elucidation using computational tools and molecular docking. Biology, 14(5), 511. https://doi.org/10.3390/biology14050511 DOI: https://doi.org/10.3390/biology14050511
Shi, M., Ahtesh, F., Mathai, M., McAinch, A. J., & Su, X. Q. (2017). Effects of fermentation conditions on the potential anti‐hypertensive peptides released from yogurt fermented by Lactobacillus helveticus and Flavourzyme ®. International Journal of Food Science & Technology, 52(1), 137–145. https://doi.org/10.1111/ijfs.13253 DOI: https://doi.org/10.1111/ijfs.13253
Suaste, N. I. J., Chel-Guerrero, L., Betancur- Ancona, D., & Bustillos, R. Z. (2023). Antioxidant and inhibitory activities of α-amylase, α-glucosidase and angiotensin-I of protein hydrolysates from “Sac-Beh” quality protein maize (QPM). Emirates Journal of Food and Agriculture. https://doi.org/10.9755/ejfa.2023.3107 DOI: https://doi.org/10.9755/ejfa.2023.3107
Sukkhown, P., Pirak, T., Jangchud, K., & Prinyawiwatkul, W. (2021). Novel peptides from dried squid head by‐products obtained from snack process. International Journal of Food Science & Technology, 56(11), 5506–5517. https://doi.org/10.1111/ijfs.15085 DOI: https://doi.org/10.1111/ijfs.15085
Sun, P., Du, W., Zheng, Y., Sun, Q., Wu, X., Zhao, Q., Mu, G., Ma, C., Li, Z., & Kong, F. (2025). Effects of Alcalase and Flavourzyme from different manufacturers on the antigenicity and mechanisms of β-lactoglobulin. LWT, 232, 118276. https://doi.org/10.1016/j.lwt.2025.118276 DOI: https://doi.org/10.1016/j.lwt.2025.118276
Tang, Y., Debnath, T., Choi, E.-J., Kim, Y. W., Ryu, J. P., Jang, S., Chung, S. U., Choi, Y.-J., & Kim, E.-K. (2018). Changes in the amino acid profiles and free radical scavenging activities of Tenebrio molitor larvae following enzymatic hydrolysis. PLOS ONE, 13(5), e0196218. https://doi.org/10.1371/journal.pone.0196218 DOI: https://doi.org/10.1371/journal.pone.0196218
Tejasari, T., Aulia, F., & Agustina, N. (2021). Inhibiting activity of angiotensin converting enzyme-1 by bean protein hydrolysate genus Phaseolus. Jurnal Penelitian Pascapanen Pertanian, 18(2), 75. https://doi.org/10.21082/jpasca.v18n2.2021.75-86 DOI: https://doi.org/10.21082/jpasca.v18n2.2021.75-86
Uraipong, C., & Zhao, J. (2016). Rice bran protein hydrolysates exhibit strong in vitro α ‐amylase, β ‐glucosidase and ACE ‐inhibition activities. Journal of the Science of Food and Agriculture, 96(4), 1101–1110. https://doi.org/10.1002/jsfa.7182 DOI: https://doi.org/10.1002/jsfa.7182
Waglay, A., & Karboune, S. (2016). Enzymatic generation of peptides from potato proteins by selected proteases and characterization of their structural properties. Biotechnology Progress, 32(2), 420–429. https://doi.org/10.1002/btpr.2245 DOI: https://doi.org/10.1002/btpr.2245
Wang, D., Zhang, M., Zou, Y., Sun, Z., & Xu, W. (2018). Optimization of flavourzyme hydrolysis condition for the preparation of antioxidant peptides from duck meat using response surface methodology. The Journal of Poultry Science, 55(3), 217–223. https://doi.org/10.2141/jpsa.0160155 DOI: https://doi.org/10.2141/jpsa.0160155
Wang, L.-S., Huang, J.-C., Chen, Y.-L., Huang, M., & Zhou, G.-H. (2015). Identification and characterization of antioxidant peptides from enzymatic hydrolysates of duck meat. Journal of Agricultural and Food Chemistry, 63(13), 3437–3444. https://doi.org/10.1021/jf506120w DOI: https://doi.org/10.1021/jf506120w
Xia, X., Song, S., Zhou, T., Zhang, H., Cui, H., Zhang, F., Hayat, K., Zhang, X., & Ho, C.-T. (2023). Preparation of saltiness-enhancing enzymatic hydrolyzed pea protein and identification of the functional small peptides of salt reduction. Journal of Agricultural and Food Chemistry, 71(21), 8140–8149. https://doi.org/10.1021/acs.jafc.3c02046 DOI: https://doi.org/10.1021/acs.jafc.3c02046
Yao, H., Yang, J., Zhan, J., Lu, Q., Su, M., & Jiang, Y. (2021). Preparation, amino acid composition, and in vitro antioxidant activity of okra seed meal protein hydrolysates. Food Science & Nutrition, 9(6), 3059–3070. https://doi.org/10.1002/fsn3.2263 DOI: https://doi.org/10.1002/fsn3.2263
Zhang, H., Zhang, Y., Javed, M., Cheng, M., Xiong, S., & Liu, Y. (2022). Gelatin hydrolysates from sliver carp (Hypophthalmichthys molitrix) improve the antioxidant and cryoprotective properties of unwashed frozen fish mince. International Journal of Food Science & Technology, 57(5), 2619–2627. https://doi.org/10.1111/ijfs.15121 DOI: https://doi.org/10.1111/ijfs.15121
Zhang, M., Zhu, L., Wu, G., Liu, T., Qi, X., & Zhang, H. (2022). Rapid screening of novel dipeptidyl peptidase-4 inhibitory peptides from pea (Pisum sativum L.) protein using peptidomics and molecular docking. Journal of Agricultural and Food Chemistry, 70(33), 10221–10228. https://doi.org/10.1021/acs.jafc.2c03949 DOI: https://doi.org/10.1021/acs.jafc.2c03949
Zhang, Y., Ke, H., Bai, T., Chen, C., Guo, T., Mu, Y., Li, H., Liao, W., Pan, Z., & Zhao, L. (2021). Characterization of umami compounds in bone meal hydrolysate. Journal of Food Science, 86(6), 2264–2275. https://doi.org/10.1111/1750-3841.15751 DOI: https://doi.org/10.1111/1750-3841.15751
Zhao, X., Cheng, Y., Xu, J., & Cui, C. (2025). Dopamine-modified hydrated silica immobilized Flavourzyme for efficient bioactive peptide release from β-lactoglobulin. Food Chemistry, 487, 144725. https://doi.org/10.1016/j.foodchem.2025.144725 DOI: https://doi.org/10.1016/j.foodchem.2025.144725
Zheng, Y., Wang, X., Zhuang, Y., Li, Y., Shi, P., Tian, H., Li, X., & Chen, X. (2020). Isolation of novel ACE‐inhibitory peptide from naked oat globulin hydrolysates in silico approach: Molecular docking, in vivo antihypertension and effects on renin and intracellular endothelin‐1. Journal of Food Science, 85(4), 1328–1337. https://doi.org/10.1111/1750-3841.15115 DOI: https://doi.org/10.1111/1750-3841.15115
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Yair Noé Ávila-Vargas, Laura García-Curiel, Jesús Guadalupe Pérez-Flores, Gabriela Mariana Rodríguez-Serrano, Elizabeth Contreras-López, Luis Guillermo González-Olivares, Rita Paz-Samaniego, Emmanuel Pérez-Escalante

This work is licensed under a Creative Commons Attribution 4.0 International License.
This journal publishes its Open Access articles under a Creative Commons license (CC BY 4.0).
You are free to:
Share — copy and redistribute the material in any medium or format for any purpose, even commercially.
Adapt — remix, transform, and build upon the material for any purpose, even commercially.
The licensor cannot revoke these freedoms as long as you follow the license terms.
Under the following terms:
Attribution — You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.
Notices:
You do not have to comply with the license for elements of the material in the public domain or where your use is permitted by an applicable exception or limitation.
No warranties are given. The license may not give you all of the permissions necessary for your intended use. For example, other rights such as publicity, privacy, or moral rights may limit how you use the material.




