Bactérias lácticas na fermentação do trigo: uma análise bibliométrica da produção de peptídeos antioxidantes
DOI:
https://doi.org/10.58951/fstoday.2025.005Palavras-chave:
Bactérias do ácido láctico, Fermentação, Produção de peptídeos antioxidantes, Hidrólise de proteína de trigo, Ingredientes alimentares funcionaisResumo
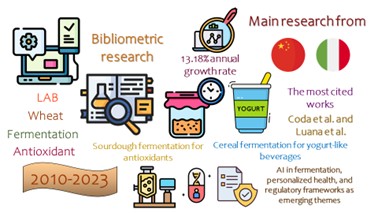
A crescente demanda por alimentos funcionais levou a um interesse em processos de fermentação que aumentam a produção de peptídeos antioxidantes. Este estudo teve como objetivo realizar uma análise bibliométrica de bactérias do ácido láctico (BAL) usadas na fermentação do trigo para produzir peptídeos antioxidantes. Para isso, dados de 2010 a 2023 foram coletados do banco de dados Web of Science®, com foco em publicações relacionadas a BAL, proteínas do trigo e peptídeos antioxidantes. Os resultados da pesquisa foram analisados usando o pacote bibliometrix R, que permitiu um exame aprofundado de tendências de pesquisa, colaborações e estudos influentes. Os resultados indicaram aumento da atividade de pesquisa, com uma taxa média de crescimento anual de 13,18% e contribuições de países como China e Itália. As principais áreas de foco incluíram métodos de fermentação, produção de peptídeos e aplicações de compostos bioativos relacionadas à saúde. As redes de colaboração destacaram a importância de parcerias internacionais no avanço deste campo. Os resultados forneceram insights sobre o desenvolvimento e a aplicação de BAL na ciência dos alimentos, enfatizando o potencial dos peptídeos bioativos produzidos a partir do trigo na promoção da saúde por meio da atividade antioxidante. Este estudo contribui para a compreensão do cenário de pesquisa e do potencial futuro do uso de LAB para produzir ingredientes alimentares funcionais.
Referências
Abera, T., Legesse, Y., Mummed, B., & Urga, B. (2016). Bacteriological quality of raw camel milk along the market value chain in Fafen zone, Ethiopian Somali regional state. BMC Research Notes, 9(1), 285. https://doi.org/10.1186/s13104-016-2088-1
Abunde, N. F., Asiedu, N. Y., & Addo, A. (2019). Modeling, simulation and optimal control strategy for batch fermentation processes. International Journal of Industrial Chemistry, 10(1), 67–76. https://doi.org/10.1007/s40090-019-0172-9
Adeyeye, O. J., & Akanbi, I. (2024). Artificial Intelligence for systems engineering complexity: A review on the use of AI and machine learning algorithms. Computer Science & IT Research Journal, 5(4), 787–808. https://doi.org/10.51594/csitrj.v5i4.1026
Akanni, G. B., & Adebo, O. A. (2024). Metabolite perturbations in fermented legumes as elucidated using metabolomics: A review. International Journal of Food Science & Technology, 59(6), 4234–4250. https://doi.org/10.1111/ijfs.17122
Akhmetsadykova, S., Baubekova, A., Konuspayeva, G., Akhmetsadykov, N., & Loiseau, G. (2014). Microflora identification of fresh and fermented camel milk from Kazakhstan. Emirates Journal of Food and Agriculture, 26(4), 327. https://doi.org/10.9755/ejfa.v26i4.17641
Alhaj, O. A., Metwalli, A. A., Ismail, E. A., Ali, H. S., Al‐Khalifa, A. S., & Kanekanian, A. D. (2018). Angiotensin converting enzyme‐inhibitory activity and antimicrobial effect of fermented camel milk (Camelus dromedarius). International Journal of Dairy Technology, 71(1), 27–35. https://doi.org/10.1111/1471-0307.12383
Alibašić, H. (2023). Developing an ethical framework for responsible artificial intelligence (AI) and machine learning (ML) applications in cryptocurrency trading: A consequentialism ethics analysis. FinTech, 2(3), 430–443. https://doi.org/10.3390/fintech2030024
Althnaian, T., Albokhadaim, I., & El-Bahr, S. M. (2013). Biochemical and histopathological study in rats intoxicated with carbontetrachloride and treated with camel milk. SpringerPlus, 2(1), 57. https://doi.org/10.1186/2193-1801-2-57
Amadou, I., Le, G.-W., Shi, Y.-H., Gbadamosi, O. S., Kamara, M. T., & Jin, S. (2011). Optimized Lactobacillus plantarum Lp6 solid-state fermentation and proteolytic hydrolysis improve some nutritional attributes of soybean protein meal: Optimized hydrolysis of fermented soybean meal. Journal of Food Biochemistry, 35(6), 1686–1694. https://doi.org/10.1111/j.1745-4514.2010.00493.x
Amigo, L., & Hernández-Ledesma, B. (2020). Current evidence on the bioavailability of food bioactive peptides. Molecules, 25(19), 4479. https://doi.org/10.3390/molecules25194479
Arcales, J. A. A., & Alolod, G. A. L. (2018). Isolation and characterization of lactic acid bacteria in philippine fermented milkfish chanos chanos-rice mixture (Burong bangus). Current Research in Nutrition and Food Science Journal, 6(2), 500–508. https://doi.org/10.12944/CRNFSJ.6.2.24
Ayyash, M., Johnson, S. K., Liu, S.-Q., Al-Mheiri, A., & Abushelaibi, A. (2018). Cytotoxicity, antihypertensive, antidiabetic and antioxidant activities of solid-state fermented lupin, quinoa and wheat by Bifidobacterium species: In-vitro investigations. LWT, 95, 295–302. https://doi.org/10.1016/j.lwt.2018.04.099
Ayyash, M., Johnson, S. K., Liu, S.-Q., Mesmari, N., Dahmani, S., Al Dhaheri, A. S., & Kizhakkayil, J. (2019). In vitro investigation of bioactivities of solid-state fermented lupin, quinoa and wheat using Lactobacillus spp. Food Chemistry, 275, 50–58. https://doi.org/10.1016/j.foodchem.2018.09.031
Babini, E., Tagliazucchi, D., Martini, S., Dei Più, L., & Gianotti, A. (2017). LC-ESI-QTOF-MS identification of novel antioxidant peptides obtained by enzymatic and microbial hydrolysis of vegetable proteins. Food Chemistry, 228, 186–196. https://doi.org/10.1016/j.foodchem.2017.01.143
Ballet, N., Renaud, S., Roume, H., George, F., Vandekerckove, P., Boyer, M., & Durand-Dubief, M. (2023). Saccharomyces cerevisiae: Multifaceted applications in one health and the achievement of sustainable development goals. Encyclopedia, 3(2), 602–613. https://doi.org/10.3390/encyclopedia3020043
Barak, S., Mudgil, D., & Khatkar, B. S. (2015). Biochemical and functional properties of wheat gliadins: A review. Critical Reviews in Food Science and Nutrition, 55(3), 357–368. https://doi.org/10.1080/10408398.2012.654863
Barathikannan, K., Tyagi, A., Shan, L., Kim, N.-H., Lee, D.-S., Park, J.-S., Chelliah, R., & Oh, D.-H. (2023). Antiobesity and antioxidative effect of fermented brown rice using in vitro with in vivo Caenorhabditis elegans model. Life, 13(2), 374. https://doi.org/10.3390/life13020374
Bento, C. B. P., De Azevedo, A. C., Detmann, E., & Mantovani, H. C. (2015). Biochemical and genetic diversity of carbohydrate-fermenting and obligate amino acid-fermenting hyper-ammonia-producing bacteria from Nellore steers fed tropical forages and supplemented with casein. BMC Microbiology, 15(1), 28. https://doi.org/10.1186/s12866-015-0369-9
Bhardwaj, G., & Singh, B. (2016). Potential MIC of bioactive peptides from fermented bovine milk to inhibit bacterial pathogens. International Journal of Advanced Multidisciplinary Research, 3(10), 30–36. https://doi.org/10.22192/ijamr.2016.03.10.004
Bi, H., Zhao, H., Lu, F., Zhang, C., Bie, X., & Lu, Z. (2015). Improvement of the nutritional quality and fibrinolytic enzyme activity of soybean meal by fermentation of B acillus subtilis: Improving the nutritional quality of soybean meal. Journal of Food Processing and Preservation, 39(6), 1235–1242. https://doi.org/10.1111/jfpp.12340
Bindelle, J., Pieper, R., Montoya, C. A., Van Kessel, A. G., & Leterme, P. (2011). Nonstarch polysaccharide-degrading enzymes alter the microbial community and the fermentation patterns of barley cultivars and wheat products in an in vitro model of the porcine gastrointestinal tract: NSP-degrading enzymes alter intestinal fermentation patterns. FEMS Microbiology Ecology, 76(3), 553–563. https://doi.org/10.1111/j.1574-6941.2011.01074.x
Bouglé, D., & Bouhallab, S. (2017). Dietary bioactive peptides: Human studies. Critical Reviews in Food Science and Nutrition, 57(2), 335–343. https://doi.org/10.1080/10408398.2013.873766
Bragason, E., Berhe, T., Dashe, D., Sørensen, K. I., Guya, M. E., & Hansen, E. B. (2020). Antimicrobial activity of novel Lactococcus lactis strains against Salmonella typhimurium DT12, Escherichia coli O157:H7 VT− and Klebsiella pneumoniae in raw and pasteurised camel milk. International Dairy Journal, 111, 104832. https://doi.org/10.1016/j.idairyj.2020.104832
C. Borresen, E., J. Henderson, A., Kumar, A., L. Weir, T., & P. Ryan, E. (2012). Fermented foods: Patented approaches and formulations for nutritional supplementation and health promotion. Recent Patents on Food, Nutrition & Agriculture, 4(2), 134–140. https://doi.org/10.2174/2212798411204020134
Chakrabarti, S., Guha, S., & Majumder, K. (2018). Food-derived bioactive peptides in human health: challenges and opportunities. Nutrients, 10(11), 1738. https://doi.org/10.3390/nu10111738
Chen, H., Mao, Y., Du, G., Li, X., Liu, Z., & Kou, J. (2020). The optimization of peptides – producing conditions for antioxidant peptides in goat milk by Lactobacillus casei L61. Acta Scientiarum Polonorum Technologia Alimentaria, 19(2), 169–176. https://doi.org/10.17306/J.AFS.0753
Chin, Y. L., Chai, K. F., & Chen, W. N. (2022). Upcycling of brewers’ spent grains via solid-state fermentation for the production of protein hydrolysates with antioxidant and techno-functional properties. Food Chemistry: X, 13, 100184. https://doi.org/10.1016/j.fochx.2021.100184
Cho, K., Beom, H.-R., Jang, Y.-R., Altenbach, S. B., Vensel, W. H., Simon-Buss, A., Lim, S.-H., Kim, M. G., & Lee, J.-Y. (2018). Proteomic profiling and epitope analysis of the complex α-, γ-, and ω-gliadin families in a commercial bread wheat. Frontiers in Plant Science, 9, 818. https://doi.org/10.3389/fpls.2018.00818
Coda, R., Rizzello, C. G., Curiel, J. A., Poutanen, K., & Katina, K. (2014). Effect of bioprocessing and particle size on the nutritional properties of wheat bran fractions. Innovative Food Science & Emerging Technologies, 25, 19–27. https://doi.org/10.1016/j.ifset.2013.11.012
Coda, R., Rizzello, C. G., Pinto, D., & Gobbetti, M. (2012). Selected lactic acid bacteria synthesize antioxidant peptides during sourdough fermentation of cereal flours. Applied and Environmental Microbiology, 78(4), 1087–1096. https://doi.org/10.1128/AEM.06837-11
De Montijo-Prieto, S., Razola-Díaz, M. D. C., Barbieri, F., Tabanelli, G., Gardini, F., Jiménez-Valera, M., Ruiz-Bravo, A., Verardo, V., & Gómez-Caravaca, A. M. (2023). Impact of lactic acid bacteria fermentation on phenolic compounds and antioxidant activity of avocado leaf extracts. Antioxidants, 12(2), 298. https://doi.org/10.3390/antiox12020298
Dong, H., Wang, H., Fu, S., & Zhang, D. (2022). CRISPR/Cas tools for enhancing the biopreservation ability of lactic acid bacteria in aquatic products. Frontiers in Bioengineering and Biotechnology, 10, 1114588. https://doi.org/10.3389/fbioe.2022.1114588
Farias, T. C., De Souza, T. S. P., Fai, A. E. C., & Koblitz, M. G. B. (2022). Critical review for the production of antidiabetic peptides by a bibliometric approach. Nutrients, 14(20), 4275. https://doi.org/10.3390/nu14204275
Farooque, S., Rose, P. M., Benohoud, M., Blackburn, R. S., & Rayner, C. M. (2018). Enhancing the potential exploitation of food waste: extraction, purification, and characterization of renewable specialty chemicals from blackcurrants (Ribes nigrum L.). Journal of Agricultural and Food Chemistry, 66(46), 12265–12273. https://doi.org/10.1021/acs.jafc.8b04373
Fayemi, O. E., & Buys, E. M. (2017). Effect of Lactobacillus plantarum on the survival of acid‐tolerant non‐O157 Shiga toxin‐producing E. coli (STEC) strains in fermented goat’s milk. International Journal of Dairy Technology, 70(3), 399–406. https://doi.org/10.1111/1471-0307.12340
Felt, U., Igelsböck, J., Schikowitz, A., & Völker, T. (2016). Transdisciplinary sustainability research in practice: Between imaginaries of collective experimentation and entrenched academic value orders. Science, Technology, & Human Values, 41(4), 732–761. https://doi.org/10.1177/0162243915626989
Feng, Y., Zhang, H., Fu, B., Iftikhar, M., Liu, G., & Wang, J. (2021). Interactions between dietary fiber and ferulic acid change the aggregation of glutenin, gliadin and glutenin macropolymer in wheat flour system. Journal of the Science of Food and Agriculture, 101(5), 1979–1988. https://doi.org/10.1002/jsfa.10814
Francisco, B. (2024). Microbial ecology and fermentation dynamics of traditional and novel fermented yogurt in Brazil. International Journal of Food Sciences, 7(2), 33–43. https://doi.org/10.47604/ijf.2596
Ganzorig, K., Urashima, T., & Fukuda, K. (2020). Exploring potential bioactive peptides in fermented bactrian camel’s milk and mare’s milk made by mongolian nomads. Foods, 9(12), 1817. https://doi.org/10.3390/foods9121817
García-Curiel, L., Pérez-Flores, J. G., González-Olivares, L. G., Guerrero-Solano, J. A., Contreras-López, E., Pérez-Escalante, E., Portillo-Torres, L. A., & Sebastián-Nicolás, J. L. (2024). Probiotics and Metabolic Syndrome: A bibliometric analysis and overview of dietary interventions. In weight loss—A multidisciplinary perspective. IntechOpen. https://doi.org/10.5772/intechopen.1004605
Garofalo, C., Norici, A., Mollo, L., Osimani, A., & Aquilanti, L. (2022). Fermentation of microalgal biomass for innovative food production. Microorganisms, 10(10), 2069. https://doi.org/10.3390/microorganisms10102069
Gille, D., Schmid, A., Walther, B., & Vergères, G. (2018). Fermented food and non-communicable chronic diseases: A review. Nutrients, 10(4), 448. https://doi.org/10.3390/nu10040448
Gojković Cvjetković, V., Marjanović‐Balaban, Ž., Vujadinović, D., Vukić, M., & Rajić, D. (2022). Investigation of the effect of cold atmospheric plasma on gliadins and glutenins extracted from wheat flour samples. Journal of Food Processing and Preservation, 46(10). https://doi.org/10.1111/jfpp.15789
Gonzales, R. R., Shintani, T., Sunami, S., Sasaki, Y., Nakagawa, K., Yoshioka, T., & Matsuyama, H. (2022). Monoamine‐modified thin film composite nanofiltration membrane for permselective separation of fermentation bioproducts. Journal of Applied Polymer Science, 139(26), e52460. https://doi.org/10.1002/app.52460
Gonzalez Viejo, C., & Fuentes, S. (2020a). Beer aroma and quality traits assessment using artificial intelligence. Fermentation, 6(2), 56. https://doi.org/10.3390/fermentation6020056
Gonzalez Viejo, C., & Fuentes, S. (2020b). Low-cost methods to assess beer quality using artificial intelligence involving robotics, an electronic nose, and machine learning. Fermentation, 6(4), 104. https://doi.org/10.3390/fermentation6040104
Gugel, I., Marchetti, F., Costa, S., Gugel, I., Baldini, E., Vertuani, S., & Manfredini, S. (2024). 2G-lactic acid from olive oil supply chain waste: Olive leaves upcycling via Lactobacillus casei fermentation. Applied Microbiology and Biotechnology, 108(1), 379. https://doi.org/10.1007/s00253-024-13217-z
Harishbhai Tilala, M., Kumar Chenchala, P., Choppadandi, A., Kaur, J., Naguri, S., Saoji, R., & Devaguptapu, B. (2024). Ethical considerations in the use of artificial intelligence and machine learning in health care: A comprehensive review. Cureus, 16(6), e62443 https://doi.org/10.7759/cureus.62443
Hasan, M. N., Sultan, M. Z., & Mar-E-Um, M. (2014). Significance of fermented food in nutrition and food science. Journal of Scientific Research, 6(2), 373–386. https://doi.org/10.3329/jsr.v6i2.16530
Hayek, S. A., & Ibrahim, S. A. (2013). Current limitations and challenges with lactic acid bacteria: A review. Food and Nutrition Sciences, 4(11), 73–87. https://doi.org/10.4236/fns.2013.411A010
Hughes, S. R., López-Núñez, J. C., Jones, M. A., Moser, B. R., Cox, E. J., Lindquist, M., Galindo-Leva, L. Á., Riaño-Herrera, N. M., Rodriguez-Valencia, N., Gast, F., Cedeño, D. L., Tasaki, K., Brown, R. C., Darzins, A., & Brunner, L. (2014). Sustainable conversion of coffee and other crop wastes to biofuels and bioproducts using coupled biochemical and thermochemical processes in a multi-stage biorefinery concept. Applied Microbiology and Biotechnology, 98(20), 8413–8431. https://doi.org/10.1007/s00253-014-5991-1
Ibrahim, S. A., Yeboah, P. J., Ayivi, R. D., Eddin, A. S., Wijemanna, N. D., Paidari, S., & Bakhshayesh, R. V. (2023). A review and comparative perspective on health benefits of probiotic and fermented foods. International Journal of Food Science & Technology, 58(10), 4948–4964. https://doi.org/10.1111/ijfs.16619
Ishikawa, D., Homma, A., Uchiyama, T., Zhao, J., & Fujii, T. (2023). Kinetic Analysis of the Degradation Reaction of β-Lactoglobulin Using Hot-Compressed Water. ACS Food Science & Technology, 3(9), 1471–1475. https://doi.org/10.1021/acsfoodscitech.3c00151
Islas-Martínez, D., Ávila-Vargas, Y. N., Rodríguez-Serrano, G. M., González-Olivares, L. G., Pérez-Flores, J. G., Contreras-López, E., Olloqui, E. J., & Pérez-Escalante, E. (2023). Multi-Bioactive Potential of a Rye Protein Isolate Hydrolysate by Enzymatic Processes. Biology Life Sciences Forum, 26(1), 38. https://doi.org/10.3390/Foods2023-15037
Jackowski, M., Niedźwiecki, Ł., Jagiełło, K., Uchańska, O., & Trusek, A. (2020). Brewer’s Spent Grains—Valuable Beer Industry By-Product. Biomolecules, 10(12), 1669. https://doi.org/10.3390/biom10121669
Kim, S.-J., Ha, S., Dang, Y.-M., Chang, J. Y., Mun, S. Y., & Ha, J.-H. (2024). Combined Non-Thermal Microbial Inactivation Techniques to Enhance the Effectiveness of Starter Cultures for Kimchi Fermentation. Journal of Microbiology and Biotechnology, 34(3), 622–633. https://doi.org/10.4014/jmb.2310.10010
Kinayang, P. G., Bachruddin, Z., & Kurniawati, A. (2021). Lactic Acid Bacteria Fermentation of High Protein Feeds: The Effect of Molasses and Incubation Time on Improving Digestibility. In 10th International Seminar and 12th Congress of Indonesian Society for Microbiology (ISISM 2019) (pp. 158-163). Atlantis Press. https://doi.org/10.2991/absr.k.210810.029
Klis, V., Pühn, E., Jerschow, J. J., Fraatz, M. A., & Zorn, H. (2023). Fermentation of Cocoa (Theobroma cacao L.) Pulp by Laetiporus persicinus Yields a Novel Beverage with Tropical Aroma. Fermentation, 9(6), 533. https://doi.org/10.3390/fermentation9060533
Kobayashi, R., Arikawa, K., Ichikawa, K., Taguchi, C., Utsunomiya, T., Iijima, M., Uchiyama, T., Kamachi, K., Nasu, I., Fukumoto, M., Kawai, Y., & Ochiai, T. (2013). Traditional Japanese Fermented Food Natto Enhances NK Cell Activity in Intestine. International Journal of Oral-Medical Sciences, 12(2), 90–94. https://doi.org/10.5466/ijoms.12.90
Konstantinidis, D., Pereira, F., Geissen, E., Grkovska, K., Kafkia, E., Jouhten, P., Kim, Y., Devendran, S., Zimmermann, M., & Patil, K. R. (2021). Adaptive laboratory evolution of microbial co‐cultures for improved metabolite secretion. Molecular Systems Biology, 17(8), e10189. https://doi.org/10.15252/msb.202010189
Kresnowati, M. T. A. P., & Febriami, H. (2015). Mapping the Effects of Starter Culture Addition on Cocoa Bean Fermentation. ASEAN Engineering Journal, 5(1), 25–37. https://doi.org/10.11113/aej.v5.15465
Lanterman, J. L., & Blithe, S. J. (2019). Benefits, Challenges, and Disincentives of Interdisciplinary Collaboration. Commoning Ethnography, 2(1), 149–165. https://doi.org/10.26686/ce.v2i1.5399
LaPensee, E., & Doshi, A. (2020). Collective creativity: Strategies for catalyzing interdisciplinary research. Journal of Science Communication, 19(04), C05. https://doi.org/10.22323/2.19040305
Leroy, F., & De Vuyst, L. (2004). Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends in Food Science & Technology, 15(2), 67–78. https://doi.org/10.1016/j.tifs.2003.09.004
Liu, F., Chen, Z., Shao, J., Wang, C., & Zhan, C. (2017). Effect of fermentation on the peptide content, phenolics and antioxidant activity of defatted wheat germ. Food Bioscience, 20, 141–148. https://doi.org/10.1016/j.fbio.2017.10.002
López-Pedrouso, M., Lorenzo, J. M., Borrajo, P., & Franco, D. (2022). In Search of Antioxidant Peptides from Porcine Liver Hydrolysates Using Analytical and Peptidomic Approach. Antioxidants, 11(1), 27. https://doi.org/10.3390/antiox11010027
Lorenzetti, L., Jacobsen, M., Lorenzetti, D. L., Nowell, L., Pethrick, H., Clancy, T., Freeman, G. (Gina), & Oddone Paolucci, E. (2022). Fostering Learning and Reciprocity in Interdisciplinary Research. Small Group Research, 53(5), 755–777. https://doi.org/10.1177/10464964221089836
Lorenzo, J. M., Munekata, P. E. S., Gómez, B., Barba, F. J., Mora, L., Pérez-Santaescolástica, C., & Toldrá, F. (2018). Bioactive peptides as natural antioxidants in food products – A review. Trends in Food Science & Technology, 79, 136–147. https://doi.org/10.1016/j.tifs.2018.07.003
Luana, N., Rossana, C., Curiel, J. A., Kaisa, P., Marco, G., & Rizzello, C. G. (2014). Manufacture and characterization of a yogurt-like beverage made with oat flakes fermented by selected lactic acid bacteria. International Journal of Food Microbiology, 185, 17–26. https://doi.org/10.1016/j.ijfoodmicro.2014.05.004
Lynch, K. M., Zannini, E., Coffey, A., & Arendt, E. K. (2018). Lactic Acid Bacteria Exopolysaccharides in Foods and Beverages: Isolation, Properties, Characterization, and Health Benefits. Annual Review of Food Science and Technology, 9(1), 155–176. https://doi.org/10.1146/annurev-food-030117-012537
Mackey, J. D., & McAllister, C. P. (2022). Musing about Interdisciplinary Research: Is Interdisciplinary Research Amusing or Bemusing? Group & Organization Management, 47(5), 899–906. https://doi.org/10.1177/10596011221093942
Magama, P., Chiyanzu, I., & Mulopo, J. (2021). Investigating Dark Fermentation as a Sustainable Organic Waste Management Technology for Producing Biohydrogen From Fruit and Vegetable Waste. https://doi.org/10.21203/rs.3.rs-955255/v1
Mandhania, M. H., Paul, D., Suryavanshi, M. V., Sharma, L., Chowdhury, S., Diwanay, S. S., Diwanay, S. S., Shouche, Y. S., & Patole, M. S. (2019). Diversity and Succession of Microbiota during Fermentation of the Traditional Indian Food Idli. Applied and Environmental Microbiology, 85(13), e00368-19. https://doi.org/10.1128/AEM.00368-19
Martirosyan, D., & Alvarado, A. (2023). Functional Foods Regulation System: Proposed Regulatory Paradigm by Functional Food Center. Functional Food Science, 3(11), 275. https://doi.org/10.31989/ffs.v3i11.1265
Martirosyan, D., Adany, A., & Kanya, H. (2021). Japan’s health food industry: An analysis of the efficacy of the FOSHU system. Bioactive Compounds in Health and Disease, 4(4), 63. https://doi.org/10.31989/bchd.v4i4.795
Marwati, T., Kurniawan, F. I., Karisma, V. L. K., Hatmi, R. U., Fajariyah, A., Fitrotin, U., Djaafar, T. F., Wikandari, R., & Rahayu, E. S. (2024). Indigenous lactic acid bacteria as a biological control agent to prevent fungi contamination in the fermentation of cocoa beans. Food Science and Technology, 44. https://doi.org/10.5327/fst.17923
Moslehishad, M., Mirdamadi, S., Ehsani, M. R., Ezzatpanah, H., & Moosavi‐Movahedi, A. A. (2013). The proteolytic activity of selected lactic acid bacteria in fermenting cow’s and camel’s milk and the resultant sensory characteristics of the products. International Journal of Dairy Technology, 66(2), 279–285. https://doi.org/10.1111/1471-0307.12017
Mutlu Sirakova, S. (2023). Forgotten Stories of Yogurt: Cultivating Multispecies Wisdom. Journal of Ethnobiology, 43(3), 250–261. https://doi.org/10.1177/02780771231194779
Muynarsk, E. S. M., De Melo Pereira, G. V., Mesa, D., Thomaz-Soccol, V., Carvalho, J. C., Pagnoncelli, M. G. B., & Soccol, C. R. (2019). Draft Genome Sequence of Pediococcus acidilactici Strain LPBC161, Isolated from Mature Coffee Cherries during Natural Fermentation. Microbiology Resource Announcements, 8(16), e00332-19. https://doi.org/10.1128/MRA.00332-19
Nasrollahzadeh, A., Khomeiri, M., Mahmoudi, M., Sadeghi, A., & Ebrahimi, M. (2019). Identification and Evaluation of the Antimicrobial Potential of Strains Derived from Traditional Fermented Dairy Products of Iran as A Biological Preservative Against Listeria monocytogenes, Staphylococcus aureus, Salmonella enterica and Escherichia coli. Iranian Journal of Medical Microbiology, 13(5), 392–405. https://doi.org/10.30699/ijmm.13.5.392
Neethirajan, S. (2024). Net Zero Dairy Farming—Advancing Climate Goals with Big Data and Artificial Intelligence. Climate, 12(2), 15. https://doi.org/10.3390/cli12020015
Nilsson, A., Johansson, E., Ekström, L., & Björck, I. (2013). Effects of a Brown Beans Evening Meal on Metabolic Risk Markers and Appetite Regulating Hormones at a Subsequent Standardized Breakfast: A Randomized Cross-Over Study. PLoS ONE, 8(4), e59985. https://doi.org/10.1371/journal.pone.0059985
Ningrum, A., Wardani, D. W., Vanidia, N., Manikharda, Sarifudin, A., Kumalasari, R., Ekafitri, R., Kristanti, D., Setiaboma, W., & Munawaroh, H. S. H. (2023). Evaluation of Antioxidant Activities from a Sustainable Source of Okara Protein Hydrolysate Using Enzymatic Reaction. Molecules, 28(13), 4974. https://doi.org/10.3390/molecules28134974
Nionelli, L., Curri, N., Curiel, J. A., Di Cagno, R., Pontonio, E., Cavoski, I., Gobbetti, M., & Rizzello, C. G. (2014). Exploitation of Albanian wheat cultivars: characterization of the flours and lactic acid bacteria microbiota, and selection of starters for sourdough fermentation. Food Microbiology, 44, 96-107. https://doi.org/10.1016/j.fm.2014.05.011
Niu, L.-Y., Jiang, S.-T., & Pan, L.-J. (2013). Preparation and evaluation of antioxidant activities of peptides obtained from defatted wheat germ by fermentation. Journal of Food Science and Technology, 50(1), 53–61. https://doi.org/10.1007/s13197-011-0318-z
Nozari, A., Asadi, M., Farahmand, F., Mirsoleimani, S. H., & Koraei, P. (2022). Acid-Base Buffering Feature of Camel Versus Cow’s Milk. https://doi.org/10.21203/rs.3.rs-1386440/v1
Nwachukwu, I. D., & Aluko, R. E. (2019). Structural and functional properties of food protein-derived antioxidant peptides. Journal of Food Biochemistry, 43(1), e12761. https://doi.org/10.1111/jfbc.12761
Nyhan, L., Sahin, A. W., Schmitz, H. H., Siegel, J. B., & Arendt, E. K. (2023). Brewers’ Spent Grain: An Unprecedented Opportunity to Develop Sustainable Plant-Based Nutrition Ingredients Addressing Global Malnutrition Challenges. Journal of Agricultural and Food Chemistry, 71(28), 10543–10564. https://doi.org/10.1021/acs.jafc.3c02489
Owusu, W. A., & Marfo, S. A. (2023). Artificial Intelligence Application in Bioethanol Production. International Journal of Energy Research, 2023, 1–8. https://doi.org/10.1155/2023/7844835
Pérez-Flores, J. G., García-Curiel, L., Pérez-Escalante, E., Contreras-López, E., & Olloqui, E. J. (2024). Arabinoxylans matrixes as a potential material for drug delivery systems development—A bibliometric analysis and literature review. Heliyon, 10(3), e25445. https://doi.org/10.1016/j.heliyon.2024.e25445
Pongsetkul, J., Benjakul, S., Sampavapol, P., Osako, K., & Faithong, N. (2014). Chemical composition and physical properties of salted shrimp paste (Kapi) produced in Thailand. International Aquatic Research, 6(3), 155–166. https://doi.org/10.1007/s40071-014-0076-4
Pontieri, P., Mennini, F. S., Magni, D., Fiano, F., Scuotto, V., Papa, A., Aletta, M., & Del Giudice, L. (2022). Sustainable open innovation for the agri-food system: Sorghum as healthy food to deal with environmental challenges. British Food Journal, 124(9), 2649–2672. https://doi.org/10.1108/BFJ-07-2021-0732
Pope, H., De Frece, A., Wells, R., Borrelli, R., Ajates, R., Arnall, A., Blake, L. J., Dadios, N., Hasnain, S., Ingram, J., Reed, K., Sykes, R., Whatford, L., White, R., Collier, R., & Häsler, B. (2021). Developing a Functional Food Systems Literacy for Interdisciplinary Dynamic Learning Networks. Frontiers in Sustainable Food Systems, 5, 747627. https://doi.org/10.3389/fsufs.2021.747627
Rachwał, K., & Gustaw, K. (2024). Lactic Acid Bacteria in Sustainable Food Production. Sustainability, 16(8), 3362. https://doi.org/10.3390/su16083362
Ramesh, V., Kumar, R., Singh, R. R. B., Kaushik, J. K., & Mann, B. (2012). Comparative evaluation of selected strains of lactobacilli for the development of antioxidant activity in milk. Dairy Science & Technology, 92(2), 179–188. https://doi.org/10.1007/s13594-011-0048-z
Ramos, L., Banc, A., Louhichi, A., Pincemaille, J., Jestin, J., Fu, Z., Appavou, M.-S., Menut, P., & Morel, M.-H. (2021). Impact of the protein composition on the structure and viscoelasticity of polymer-like gluten gels. Journal of Physics: Condensed Matter, 33(14), 144001. https://doi.org/10.1088/1361-648X/abdf91
Rau, M. H., Gaspar, P., Jensen, M. L., Geppel, A., Neves, A. R., & Zeidan, A. A. (2022). Genome-Scale Metabolic Modeling Combined with Transcriptome Profiling Provides Mechanistic Understanding of Streptococcus thermophilus CH8 Metabolism. Applied and Environmental Microbiology, 88(16), e00780-22. https://doi.org/10.1128/aem.00780-22
Raveschot, C., Cudennec, B., Coutte, F., Flahaut, C., Fremont, M., Drider, D., & Dhulster, P. (2018). Production of Bioactive Peptides by Lactobacillus Species: From Gene to Application. Frontiers in Microbiology, 9, 2354. https://doi.org/10.3389/fmicb.2018.02354
Reyes-Díaz, A., Mata-Haro, V., Hernández, J., González-Córdova, A. F., Hernández-Mendoza, A., Reyes-Díaz, R., Torres-Llanez, M. J., Beltrán-Barrientos, L. M., & Vallejo-Cordoba, B. (2018). Milk Fermented by Specific Lactobacillus Strains Regulates the Serum Levels of IL-6, TNF-α and IL-10 Cytokines in a LPS-Stimulated Murine Model. Nutrients, 10(6), 691. https://doi.org/10.3390/nu10060691
Ribeiro, G. O., Rodrigues, L. D. A. P., Santos, T. B. S. D., Alves, J. P. S., Oliveira, R. S., Nery, T. B. R., Barbosa, J. D. V., & Soares, M. B. P. (2023). Innovations and developments in single cell protein: Bibliometric review and patents analysis. Frontiers in Microbiology, 13, 1093464. https://doi.org/10.3389/fmicb.2022.1093464
Rohm, H., Brennan, C., Turner, C., Günther, E., Campbell, G., Hernando, I., Struck, S., & Kontogiorgos, V. (2015). Adding Value to Fruit Processing Waste: Innovative Ways to Incorporate Fibers from Berry Pomace in Baked and Extruded Cereal-based Foods—A SUSFOOD Project. Foods, 4(4), 690–697. https://doi.org/10.3390/foods4040690
Rutherfurd-Markwick, K. J. (2012). Food proteins as a source of bioactive peptides with diverse functions. British Journal of Nutrition, 108(S2), S149–S157. https://doi.org/10.1017/S000711451200253X
Sari, K., Prihadyanti, D., & Hidayat, D. (2019). Drivers of Industry Convergence: The Case of Functional Food Industry in Indonesia. STI Policy and Management Journal, 4(1), 65-76. https://doi.org/10.14203/STIPM.2019.153
Shukla, P., Sakure, A., Maurya, R., Bishnoi, M., Kondepudi, K. K., Das, S., Liu, Z., Padhi, S., Rai, A. K., & Hati, S. (2023). Antidiabetic, angiotensin‐converting enzyme inhibitory and anti‐inflammatory activities of fermented camel milk and characterisation of novel bioactive peptides from lactic‐fermented camel milk with molecular interaction study. International Journal of Dairy Technology, 76(1), 149–167. https://doi.org/10.1111/1471-0307.12910
Tachie, C. Y. E., Onuh, J. O., & Aryee, A. N. A. (2024). Nutritional and potential health benefits of fermented food proteins. Journal of the Science of Food and Agriculture, 104(3), 1223–1233. https://doi.org/10.1002/jsfa.13001
Tadesse, S. A., & Emire, S. A. (2020). Production and processing of antioxidant bioactive peptides: A driving force for the functional food market. Heliyon, 6(8), e04765. https://doi.org/10.1016/j.heliyon.2020.e04765
Tak, L., Bais, B., Singh, R., Singh, S., & Nayak, T. C. (2018). Assessment of Probiotic and Neutraceutical Properties of Camel Milk Yoghurt. International Journal of Current Microbiology and Applied Sciences, 7(10), 3351–3357. https://doi.org/10.20546/ijcmas.2018.710.388
Torres‐Fuentes, C., Alaiz, M., & Vioque, J. (2014). Chickpea chelating peptides inhibit copper‐mediated lipid peroxidation. Journal of the Science of Food and Agriculture, 94(15), 3181–3188. https://doi.org/10.1002/jsfa.6668
Tymensen, L. D., Beauchemin, K. A., & McAllister, T. A. (2012). Structures of free-living and protozoa-associated methanogen communities in the bovine rumen differ according to comparative analysis of 16S rRNA and mcrA genes. Microbiology, 158(7), 1808–1817. https://doi.org/10.1099/mic.0.057984-0
Uno, S., Kodama, D., Yukawa, H., Shidara, H., & Akamatsu, M. (2020). Quantitative analysis of the relationship between structure and antioxidant activity of tripeptides. Journal of Peptide Science, 26(3), e3238. https://doi.org/10.1002/psc.3238
Verni, M., Pontonio, E., Krona, A., Jacob, S., Pinto, D., Rinaldi, F., Verardo, V., Díaz-de-Cerio, E., Coda, R., & Rizzello, C. G. (2020). Bioprocessing of Brewers’ Spent Grain Enhances Its Antioxidant Activity: Characterization of Phenolic Compounds and Bioactive Peptides. Frontiers in Microbiology, 11, 1831. https://doi.org/10.3389/fmicb.2020.01831
Vinayamohan, P. G., Viju, L. S., Joseph, D., & Venkitanarayanan, K. (2023). Fermented Foods as a Potential Vehicle of Antimicrobial-Resistant Bacteria and Genes. Fermentation, 9(7), 688. https://doi.org/10.3390/fermentation9070688
Wang, C., Wei, W., Wu, L., Wang, Y., Dai, X., & Ni, B.-J. (2024). A Novel Sustainable and Self-Sufficient Biotechnological Strategy for Directly Transforming Sewage Sludge into High-Value Liquid Biochemicals. Environmental Science & Technology, 58(28), 12520–12531. https://doi.org/10.1021/acs.est.4c03165
Wang, N., Xiong, Y., Wang, X., Guo, L., Lin, Y., Ni, K., & Yang, F. (2022). Effects of Lactobacillus plantarum on Fermentation Quality and Anti-Nutritional Factors of Paper Mulberry Silage. Fermentation, 8(4), 144. https://doi.org/10.3390/fermentation8040144
Wei, G., Regenstein, J. M., & Zhou, P. (2021). The fermentation‐time dependent proteolysis profile and peptidomic analysis of fermented soybean curd. Journal of Food Science, 86(8), 3422–3433. https://doi.org/10.1111/1750-3841.15823
Workie, M. (2020). Functional Genomics, Metabolic Engineering and Mutagenesis Study of Lactic Acid Bacterial Strains in Traditional Food Fermentation, Human Health and Their Potential Applications. Advances in Life Science and Technology, 83, 19. https://doi.org/10.7176/ALST/83-03
Wu, Y. Y., Liu, F. J., Li, L. H., Yang, X. Q., Deng, J. C., & Chen, S. J. (2011). Isolation and Identification of Nitrite-Degrading Lactic Acid Bacteria from Salted Fish. Advanced Materials Research, 393, 828–834. https://doi.org/10.4028/www.scientific.net/AMR.393-395.828
Xu, J., Chen, Y., Fan, X., Shi, Z., Liu, M., Zeng, X., Wu, Z., & Pan, D. (2022). Isolation, identification, and characterization of corn-derived antioxidant peptides from corn fermented milk by Limosilactobacillus fermentum. Frontiers in Nutrition, 9, 1041655. https://doi.org/10.3389/fnut.2022.1041655
Yang, H., Xing, R., Hu, L., Liu, S., & Li, P. (2016). Accumulation of γ‐aminobutyric acid by Enterococcus avium 9184 in scallop solution in a two‐stage fermentation strategy. Microbial Biotechnology, 9(4), 478–485. https://doi.org/10.1111/1751-7915.12301
Zaky, A. A., Simal-Gandara, J., Eun, J.-B., Shim, J.-H., & Abd El-Aty, A. M. (2022). Bioactivities, Applications, Safety, and Health Benefits of Bioactive Peptides From Food and By-Products: A Review. Frontiers in Nutrition, 8, 815640. https://doi.org/10.3389/fnut.2021.815640
Zhang, B., Liu, J., Liu, C., Liu, B., Yu, Y., & Zhang, T. (2020). Bifunctional peptides with antioxidant and angiotensin‐converting enzyme inhibitory activity in vitro from egg white hydrolysates. Journal of Food Biochemistry, 44(9), e13347. https://doi.org/10.1111/jfbc.13347
Zhang, H., Zhang, Z., He, D., Li, S., & Xu, Y. (2022). Optimization of Enzymatic Hydrolysis of Perilla Meal Protein for Hydrolysate with High Hydrolysis Degree and Antioxidant Activity. Molecules, 27(3), 1079. https://doi.org/10.3390/molecules27031079
Zhang, J., Caiyin, Q., Feng, W., Zhao, X., Qiao, B., Zhao, G., & Qiao, J. (2016). Enhance nisin yield via improving acid-tolerant capability of Lactococcus lactis F44. Scientific Reports, 6(1), 27973. https://doi.org/10.1038/srep27973
Zhang, O. L., Niu, J. Y., Yin, I. X., Yu, O. Y., Mei, M. L., & Chu, C. H. (2022). Growing Global Research Interest in Antimicrobial Peptides for Caries Management: A Bibliometric Analysis. Journal of Functional Biomaterials, 13(4), 210. https://doi.org/10.3390/jfb13040210
Zhang, Q., Wang, Y., Zhao, L., Su, G., Ding, W., Zheng, L., & Zhao, M. (2024). A Comparative Study of the Stability, Transport, and Structure–Activity Relationship of Round Scad Derived Peptides with Antineuroinflammatory Ability. Journal of Agricultural and Food Chemistry, 72(30), 17017–17029. https://doi.org/10.1021/acs.jafc.4c03029
Zhang, X., Li, X., Zhao, Y., Zheng, Q., Wu, Q., & Yu, Y. (2022). Nanocarrier system: An emerging strategy for bioactive peptide delivery. Frontiers in Nutrition, 9, 1050647. https://doi.org/10.3389/fnut.2022.1050647
Zhang, Y., Jiang, Z., Li, Y., Feng, Z., Zhang, X., Zhou, R., Liu, C., & Yang, L. (2022). The Combined Cultivation of Feruloyl Esterase-Producing Strains with CMCase and Xylanase-Producing Strains Increases the Release of Ferulic Acid. Microorganisms, 10(10), 1889. https://doi.org/10.3390/microorganisms10101889
Zhang, Y., Li, Y., Quan, Z., Xiao, P., & Duan, J.-A. (2024). New Insights into Antioxidant Peptides: An Overview of Efficient Screening, Evaluation Models, Molecular Mechanisms, and Applications. Antioxidants, 13(2), 203. https://doi.org/10.3390/antiox13020203
Zhao, M., Rao, J., & Chen, B. (2022). Effect of high oleic soybean oil oleogels on the properties of doughs and corresponding bakery products. Journal of the American Oil Chemists’ Society, 99(11), 1071–1083. https://doi.org/10.1002/aocs.12594
Zhao, M., Su, X. Q., Nian, B., Chen, L. J., Zhang, D. L., Duan, S. M., Wang, L. Y., Shi, X. Y., Jiang, B., Jiang, W. W., Lv, C. Y., Wang, D. P., Shi, Y., Xiao, Y., Wu, J.-L., Pan, Y. H., & Ma, Y. (2019). Integrated Meta-omics Approaches To Understand the Microbiome of Spontaneous Fermentation of Traditional Chinese Pu-erh Tea. mSystems, 4(6), e00680-19. https://doi.org/10.1128/msystems.00680-19
Zhu, Y., Lao, F., Pan, X., & Wu, J. (2022). Food Protein-Derived Antioxidant Peptides: Molecular Mechanism, Stability and Bioavailability. Biomolecules, 12(11), 1622. https://doi.org/10.3390/biom12111622
Zhu, Z., Yang, J., Huang, T., Pius Bassey, A., Huang, M., & Huang, J. (2023). The generation and application of antioxidant peptides derived from meat protein: A review. Food Science of Animal Products, 1(1), 9240005. https://doi.org/10.26599/FSAP.2023.9240005
Zibaee, S., Hosseini, S. M. A.-R., Yousefi, M., Taghipour, A., Kiani, M. A., & Noras, M. R. (2015). Nutritional and Therapeutic Characteristics of Camel Milk in Children: A Systematic Review. Electronic Physician, 7(7), 1523–1528. https://doi.org/10.19082/1523
Zou, T.-B., He, T.-P., Li, H.-B., Tang, H.-W., & Xia, E.-Q. (2016). The Structure-Activity Relationship of the Antioxidant Peptides from Natural Proteins. Molecules, 21(1), 72. https://doi.org/10.3390/molecules21010072
Zou, X., Wang, X., Li, L., Peng, P., Ma, Q., Hu, X., & Appels, R. (2022). Effects of Composition and Strength of Wheat Gluten on Starch Structure, Digestion Properties and the Underlying Mechanism. Foods, 11(21), 3432. https://doi.org/10.3390/foods112
Downloads
Publicado
Como Citar
Edição
Seção
Licença
Copyright (c) 2025 Zaira Daniela Solís-Macías, Jesús Guadalupe Pérez-Flores , Laura García-Curiel , Luis Guillermo González-Olivares , Gabriela Mariana Rodríguez-Serrano, Elizabeth Contreras-López , Alexis Ayala-Niño , Emmanuel Pérez-Escalante

Este trabalho está licenciado sob uma licença Creative Commons Attribution 4.0 International License.
Esta revista publica seus artigos em Acesso Aberto sob licença Creative Commons (CC BY 4.0).
Você é livre para:
Compartilhar — copie e redistribua o material em qualquer meio ou formato para qualquer finalidade, inclusive comercial.
Adaptar – remixar, transformar e desenvolver o material para qualquer finalidade, até mesmo comercial.
O licenciante não pode revogar essas liberdades desde que você siga os termos da licença.
Nos seguintes termos:
Atribuição — Você deve dar o devido crédito, fornecer um link para a licença e indicar se foram feitas alterações. Você pode fazê-lo de qualquer maneira razoável, mas não de forma que sugira que o licenciante endossa você ou seu uso.
Sem restrições adicionais — Você não pode aplicar termos legais ou medidas tecnológicas que restrinjam legalmente outras pessoas de fazerem qualquer coisa que a licença permita.
Avisos:
Você não precisa cumprir a licença para elementos do material de domínio público ou onde seu uso for permitido por uma exceção ou limitação aplicável.
Nenhuma garantia é dada. A licença pode não conceder todas as permissões necessárias para o uso pretendido. Por exemplo, outros direitos, como publicidade, privacidade ou direitos morais, podem limitar a forma como você utiliza o material.




